Abstract
Extracellular Vesicles (EVs) are known to play a crucial role in intracellular communication (Contreras-Naranjo et al., 2018). They are released by most cell types during apoptosis (Borges et al., 2013). However, healthy cells also secrete EVs which deliver, among others, nucleic acids (Ratajczak et al., 2006; Cai et al., 2013). After being taken up, these small fractions of RNAs and DNAs can alter the behaviour of their recipient cell (Fischer et al., 2016). The relatively small size of Extracellular Vesicles hindered the analysis of nucleic acid containing EVs to this point. Here we report an easy and quick method to analyse size and concentration of nucleic acid containing EVs by fluorescent NTA (f-NTA) after staining with DAPI.
Introduction
Analysis of nucleic acid containing EVs is of great interest as it is known that this horizontal RNA and DNA transfer orchestrates the transcription of the target cell and has therefore a strong impact on a wide range of different diseases (Sansone et al, 2017). Moreover, it has been shown that the appearance of nucleic acid containing EVs can be altered by diseases (Kitai et al., 2017).
4′,6-Diamidin-2-phenylindol (DAPI) is a well-known fluorescent stain which is used for many different applications such as fluorescence microscopy or flow cytometry. It can pass cell membranes and is therefore an ideal dye to detect nucleic acids inside intact membranous particles. DAPI binds the AT rich regions of double stranded DNA and also intercalates in RNA. Its absorption maximum is at 358nm, and when bound to DNA its emission maximum is in the blue range. When bound to RNA the emission is about 5-fold weaker and the excitation maximum is shifted into the green range.
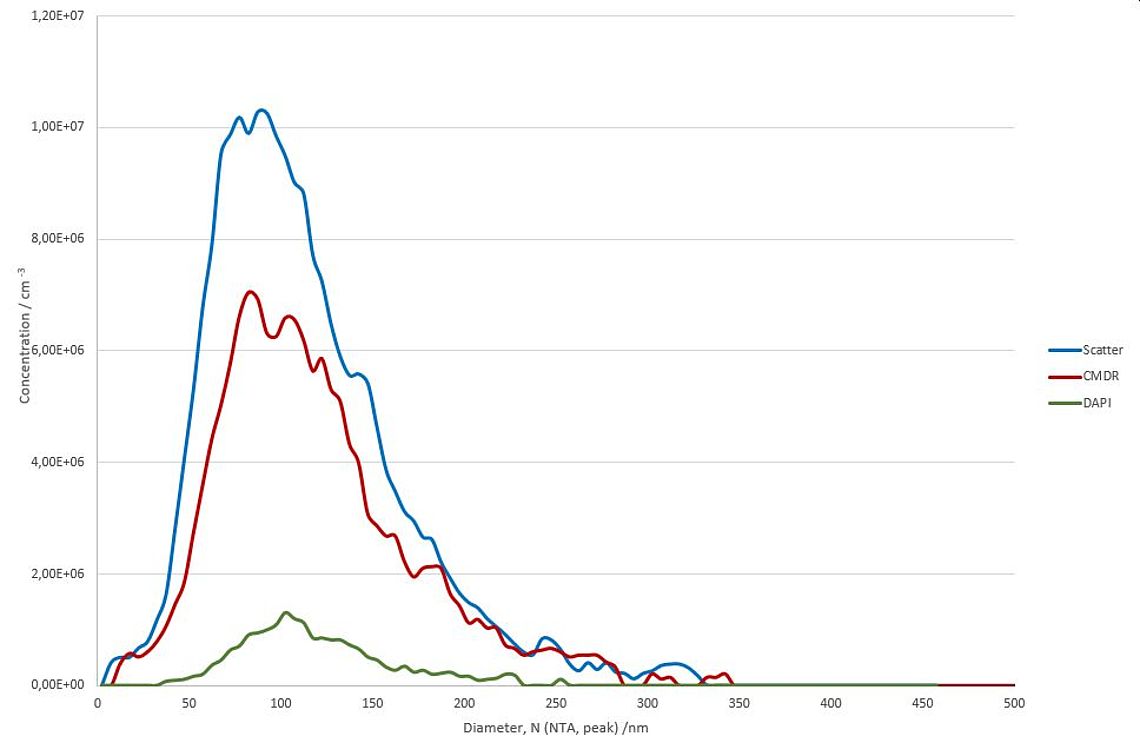
Figure 1: NTA size measurement by detection of scattered light after excitation with 488nm (red line) and fluorescence light after excitation with 640nm (red line) and 405nm (green line) of HTC116 cell line derived exosomes stained with DAPI.
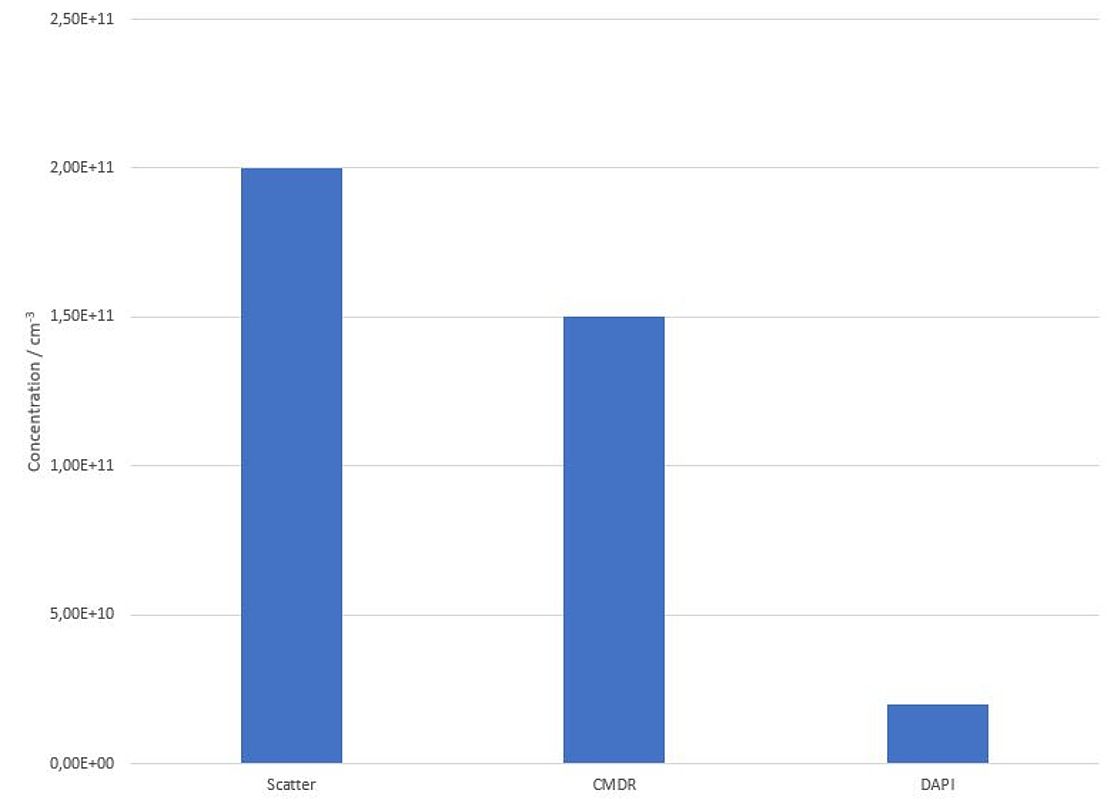
Figure 2: NTA concentration measurement by detection of scattered light and fluorescence light after excitation with 488nm (Scatter) 640nm (CMDR) and 405nm (DAPI) of HTC116 cell line derived exosomes stained with DAPI and CMDR.
The new Particle Metrix ZetaView® QUATT is equipped with a 405nm as well as with a 488nm, 520 nm and 640 nm excitation lasers and suitable long pass fluorescence filter sets making the instrument a perfect tool to analyse DNA- as well as RNA-bound DAPI.
To prove this capability, HTC116 cell line derived exosomes (HansaBioMed, Estonia) were used and simultaneously stained with DAPI and Cell Mask© Deep Red (Thermo Fisher, USA) followed by size and concentration measurements with the Particle Matrix ZetaView® QUATT.
Methods
Lyophilized exosomes derived from a HTC116 cell line (HansaBioMed, Estonia) have been resuspended with water at 1µg/µl. Staining was performed with 100µg/ml DAPI (Sigma-Aldrich, USA) and Cell Mask© Deep Red (Thermo Fisher, USA) for 2 hours at room temperature. Prior to analysis stained EVs were diluted 1:1.000 with PBS (Thermo Fisher, USA). NTA measurements were performed with a ZetaView® PMX-420 Analyser (Particle Metrix, Germany) The simultaneously built-in lasers with an excitation wavelength of 405nm and 640nm, are used for fluorescence detection whereas the 488 nm laser was used for all scatter applications. Videos were taken at 30 frames per second and analyzed for size and concentration using the ZetaView software version 8.05.10 (Particle Metrix, Germany) with anti-bleach technology.
Results & Conclusions
The results clearly show an overall scatter concentration of 2.0 x 1011 particles/ml (see Figure 2). The mean size was calculated to 106.5 nm. (Figure 1 blue curve).
Purity measurements with Cell Mask© Deep Red (CMDR) however indicate that only about 75% (concentration = 1,5 x 1011) of the total particles are surrounded by a biological membrane (see Figure 2). The size measurement of the membranous particles fully confirms the scatter measurement. The mean diameter of the CMDR positive vesicles was calculated to be 113.2 nm (see Figure 1, red curve). All other particles that are non-visible in the red fluorescence channel (CMDR) but detected in scatter mode are most likely inorganic nanobubbles, insoluble salt crystals or protein aggregates.
Internal DNA / RNA staining of the HTC116 derived EVs with DAPI was performed as described under Methods. By exciting the samples with the 405nm laser, 1 x 1010 particles/ml were detected, which is about 13% of the total membranous particles (see Figure 2). This indicates that only a fraction of the HTC116 cell line derived EVs contain detectable amount of nucleic acids. However, it remains unclear if the DAPI-negative vesicles do not contain nucleic acids or if the DNA / RNA concentration in those vesicles is below the detection limit of the ZetaView NTA System. The median size of the DAPI positive fraction tend to be a bit higher (116.9 nm) compared to the fraction of all membranous particles but is still within the statistical variance of the measurement method (Figure 1, green curve).
These results show that the ZetaView could be a very useful tool to analyse size and concentration of nucleic acid containing EVs.
References
Contreras-Naranjo J.C., Wu H.-J., and Ugaz. Microfluidics for exosome isolation and analysis: Enabling liquid biopsy for personalized medicine. Lab Chip 2018 17(21): 3558–3577
Borges F.T., Reis L.A., Schor N. Extracellular vesicles: structure, function, and potential clinical uses in renal diseases. Braz J Med Biol Res 2013 46(10): 824-30.
Ratajczak J., Miekus K., Kucia M., Zhang J., Reca R., Dvorak P., Ratajczak M.Z. Embryonic stem cell-derived microvesicles reprogram hematopoietic progenitors: evidence for horizontal transfer of mRNA and protein delivery. Leukemia 2006 20(5):847-56. DOI: 10.1038/sj.leu.2404132.
Cai J., Han Y., Ren H., Chen C., He D., Zhou L., Eisner G.M., Asico L.D., Jose P.A., Zeng C. Extracellular vesicle-mediated transfer of donor genomic DNA to recipient cells is a novel mechanism for genetic influence between cells. J Mol Cell Biol. 2013 5(4):227-38. doi: 10.1093/jmcb/mjt011
Fischer S., Cornils K., Speiseder T., Badbaran A., Reimer R., Indenbirken D., Grundhoff A., Brunswig-Spickenheier B., Alawi M., Lange C. Indication of Horizontal DNA Gene Transfer by Extracellular Vesicles. PLoS One 2016 Sep 29;11(9):e0163665.
Sansone P., Savini C., Kurelac I., Chang Q., Benedetta L., Strillacci A., Stepanova A., Iommarini L., Mastroleo C., Daly L., Galkin A., Thakur B., Soplop N., Uryu K., Hoshino A., Norton L., Bonafé M., Cricca M., Gasparre G., Lyden D., Bromberg J. Packaging and transfer of mitochondrial DNA via exosomes regulate escape from dormancy in hormonal therapy-resistant breast cancer. Proc Natl Acad Sci 2017 doi: 10.1073/pnas.1704862114
Kitai Y., Kawasaki T., Sueyoshi T., Kobiyama K., Ishii K.J., Zou J., Akira S., Matsuda T., Kawai T. DNA-Containing Exosomes Derived from Cancer Cells Treated with Topotecan Activate a STING-Dependent Pathway and Reinforce Antitumor Immunity. J Immunol. 2017 ;198(4):1649-1659.
